
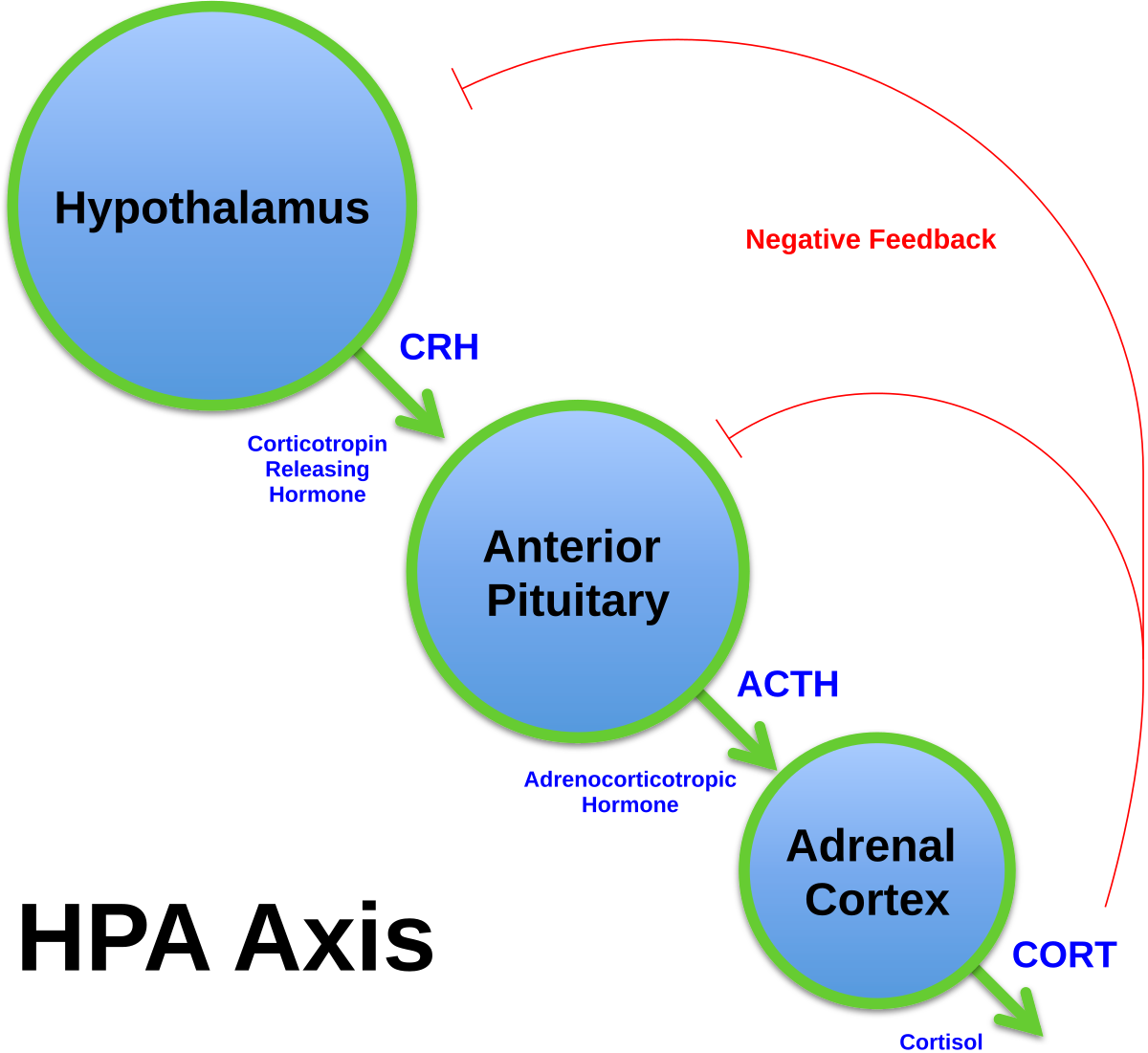


Hypothalamo–Pituitary Axis
1. Big Picture (Orientation Logic)
- The pituitary gland (hypophysis) is the master endocrine relay between the brain (hypothalamus) and the peripheral endocrine organs.
- It sits at the base of the brain, physically connected to the hypothalamus and functionally controlled by it.
- Its design reflects dual control:
- Neural control → posterior pituitary
- Endocrine (portal blood) control → anterior pituitary
2. Size, Weight & Sexual Dimorphism
- Weight: ~0.5 g in adults
- Size: 10–15 mm in each dimension
- Sex difference:
- Heavier in women
- Enlarges during pregnancy
➡️ Logic: reflects increased hormonal demand (especially prolactin).
3. Exact Location & Relations (Exam-Critical Spatial Logic)
Position
- Protrudes from the inferior surface of the hypothalamus
- Lies in sella turcica of the sphenoid bone
- Covered by diaphragma sellae (dural fold)
Key Neighbours
- Superior → optic chiasm
- Lateral → cavernous sinuses
- Superior/posterior → floor of the third ventricle
➡️ Logic:
- Optic chiasm proximity → visual field defects in pituitary tumours
- Cavernous sinus proximity → cranial nerve palsies
4. Structural Division (Why Two Lobes Exist)
The pituitary is not one organ embryologically or functionally.

A. Anterior Pituitary (Adenohypophysis)
- Glandular
- Hormone synthesis occurs here
- Subdivisions:
- Pars distalis → main hormone-secreting part
- Pars tuberalis → wraps around stalk
B. Posterior Pituitary (Neurohypophysis)
- Neural tissue
- Stores and releases hypothalamic hormones
- Components:
- Pars nervosa
- Pituitary stalk (infundibulum)
- Median eminence
➡️ Logic:
Anterior = makes hormones
Posterior = releases hormones made in hypothalamus
5. Pituitary Stalk & Hypothalamus (Connection Logic)
Hypothalamic Boundaries
- Anterior → optic chiasm
- Posterior → mammillary bodies
- Lateral → temporal lobes (sulci)
- Dorsal → thalamus (hypothalamic sulcus)
Median Eminence
- Central inferior hypothalamic region
- Highly vascular
- Contains primary capillary plexus of portal system
Connections
- Neural connections → posterior pituitary
- Endocrine (portal) connections → anterior pituitary
➡️ Logic: one stalk, two communication methods.
6. Blood Supply (High-Yield Logic Flow)
Arterial Supply
- Hypothalamus → Circle of Willis
- Anterior pituitary → Superior hypophyseal arteries
- Posterior pituitary → Inferior hypophyseal arteries
Portal System Logic (Why It Exists)
- Superior hypophyseal arteries
- → Primary capillary plexus in median eminence
- → Long portal veins (run along ventral stalk)
- → Secondary capillary plexus in anterior pituitary
- → Hormones released locally
➡️ Purpose: undiluted, rapid hypothalamic control
- Short portal veins from inferior hypophyseal system → minor contribution
- Anterior pituitary = one of the most vascular tissues in mammals
Venous Drainage
- Anterior pituitary → cavernous sinus→ superior & inferior petrosal sinuses→ jugular vein
7. Histology — Hypothalamic Neurons (3 Types)
1. Magnocellular Neurons
- Secrete:
- AVP (vasopressin)
- Oxytocin
- Cell bodies:
- Supraoptic nucleus
- Paraventricular nucleus
- Axons descend via stalk → posterior pituitary → hormone released into blood
2. Hypophysiotrophic Neurons
- Location:
- Paraventricular nucleus
- Arcuate nucleus
- Medial basal hypothalamus
- Release hormones into portal circulation
- Hormones:
- TRH
- CRH
- GHRH
- Somatostatin
- GnRH
- Dopamine
➡️ Logic: control anterior pituitary secretion.
3. Projection Neurons
- Found in:
- Paraventricular nucleus
- Arcuate nucleus
- Lateral hypothalamic area
- Project to:
- Brainstem
- Spinal cord
- Regulate autonomic nervous system
8. Pituitary Histology
Adenohypophysis
- Cell types (based on staining):
- Basophils
- Acidophils
- Chromophobes
- Staining reflects hormone-containing granules
Neurohypophysis
- Not glandular
- Composed of:
- Axon bundles from hypothalamic neurons
- Supporting glial cells
9. Embryology (Why Tumours Occur Where They Do)

Anterior Pituitary
- Origin: oral ectoderm
- Forms from Rathke pouch
- Appears at weeks 4–5
- Pouch:
- Detaches from oral cavity
- Lumen → small cleft
- Upper part → pars tuberalis
- Remnants → craniopharyngiomas
Posterior Pituitary
- Origin: neural tissue
- Downward evagination from floor of 3rd ventricle
- Lumen closes → neural stalk
- Upper recess → median eminence
Key Developmental Milestones
- Cleft of Rathke pouch → boundary between lobes
- Hypothalamo–pituitary axis established by week 20
- Hormone-secreting cell differentiation → transcription factor-driven
10. Physiology — Hypothalamic Functions (Focused Scope)
General roles:
- Autonomic nervous system control
- Thermoregulation
- Hunger & thirst
- Memory, behaviour, emotions
Endocrine focus:
- Regulates pituitary hormone secretion via:
- Neural pathways
- Portal circulation
Final Logic Lock (One-Line Integration)
The pituitary gland is a dual-origin, dual-control endocrine organ lying in the sella turcica, structurally and embryologically divided into glandular anterior and neural posterior lobes, connected to the hypothalamus by neural axons and a specialized portal circulation that allows precise central regulation of endocrine function.
Anterior Pituitary Gland — Logic-Based Physiology Note

1. Core Regulatory Logic (How the Anterior Pituitary Is Controlled)
The anterior pituitary is regulated by three interacting control systems:
- Hypothalamic input
- Releasing factors
- Inhibitory factors
- Delivered via the hypothalamo–hypophysial portal system
- Feedback from circulating peripheral hormones
- Mainly negative feedback
- Acts at:
- Pituitary level
- Hypothalamic level
- Local pituitary control
- Paracrine secretion
- Autocrine secretion
➡️ Logic: control is central + peripheral + local, allowing tight hormonal precision.
2. Major Hormones of the Anterior Pituitary (Clinical Set)
The anterior pituitary synthesises and secretes six major hormones:
- Growth hormone (GH / somatotrophin)
- Thyroid-stimulating hormone (TSH / thyrotrophin)
- Adrenocorticotrophin (ACTH)
- Follicle-stimulating hormone (FSH)
- Luteinising hormone (LH)
- Prolactin (PRL)
➡️ Note: physiological control of TSH and ACTH described elsewhere (not repeated here).
3. Growth Hormone (GH)
Cell of Origin
- Secreted by somatotrophs
- Somatotrophs = 40–50% of anterior pituitary cells
Structure
- Single-chain polypeptide
- 191 amino acids
- Structural homology with:
- Prolactin
- Human chorionic gonadotrophin (hCG)
- hCG = GH variant synthesised exclusively in placenta
Physiological Actions of GH
A. Growth & Differentiation
- Promotes:
- Linear growth
- Cell differentiation
- Tissue growth
- Important in lactation(Lactation is highly energy-demanding. GH ensures the mother can meet this demand)
B. Protein Metabolism
- ↑ Amino acid uptake
- ↑ Protein synthesis
- ↓ Protein oxidation
➡️ Net effect: anabolic
C. Fat Metabolism
- Stimulates:
- Triglyceride breakdown
- Fatty acid oxidation in adipocytes
➡️ Net effect: lipolysis
D. Carbohydrate Metabolism
- Maintains blood glucose levels by:
- ↓ Insulin-mediated glucose uptake in peripheral tissues
- ↓ Insulin-mediated glucose synthesis in liver
- Stimulates insulin secretion
- GH administration → hyperinsulinaemia
➡️ Logic: GH is diabetogenic but insulin-stimulating
4. IGF System (Indirect GH Effects)
GH acts:
- Directly
- Indirectly via insulin-like growth factors (IGFs)
IGF Family
- Insulin
- IGF-I
- IGF-II
All share:
- Structural similarities
- Metabolic roles
- Roles in cellular proliferation & differentiated tissue function
(via IGF-I receptor)
Developmental Roles
- IGF-II → major fetal growth factor
- IGF-I → key postnatal growth mediator
- Insulin → contributes to fetal growth
- Explains fetal macrosomia in maternal diabetes
5. Regulation of GH Secretion
Hypothalamic Control
- GHRH → stimulates GH
- Somatostatin → inhibits GH
GHRH
- Released from median eminence
- Travels via portal capillaries to anterior pituitary
Somatostatin
- 14-amino-acid peptide
- Synthesised mainly in anterior periventricular nuclei
Physiological Modulators of growth hormone
- Sleep, decreased at REM sleep
- Exercise
- Stress
- Blood glucose levels

Feedback Control
- IGF-I:
- Negative feedback on:
- Pituitary
- Hypothalamus
- Estradiol:
- Increases tissue sensitivity to GH
6. GH Secretion Pattern
- Begins early fetal life
- Continues throughout life
- Secretion progressively declines with age
- Pulsatile secretion
Age-Related Pattern
- High in childhood
- Peak at puberty
- Falls in adulthood
Adult Pattern
- ~5 pulses per 24 hours
- Largest pulse:
- At onset of sleep
- Half-life: ~20 minutes
7. Prolactin (PRL)
Cell of Origin
- Secreted by lactotrophs
- Lactotrophs = 10–15% of anterior pituitary cells
Structure
- Single-chain protein
- 199 amino acids
- Three disulphide bridges
- Strong structural homology with GH
- PRL and GH receptors are also structurally similar
Estrogen Effects
- Estrogen:
- Stimulates lactotroph proliferation
- Result:
- Increased lactotroph numbers in premenopausal women
- Marked increase during pregnancy
Physiological Actions
Primary Role in Humans
- Preparation of female breast for lactation
Other Sites of Action
- Gonads
- Lymphoid cells
- Liver
➡️ PRL receptors widely expressed; many functions remain unclear.
Key Clinical Observations
- Men have same PRL levels as non-lactating women
- PRL affects:
- Hypothalamo–pituitary–gonadal axis
- Inhibits pulsatile GnRH secretion
- Alters steroidogenic enzyme activity
Regulation of PRL Secretion
Unique Hypothalamic Control
- Predominantly inhibitory
(unlike all other pituitary hormones)
➡️ Damage to hypothalamus → increased PRL
Inhibitory Factors
- Dopamine (main regulator)
- Released into portal veins
- Somatostatin
Stimulatory Factors
- TRH
- Other releasing factors
- Pregnancy
- Lactation (suckling)
- Estrogen
- Opioids
- Dopamine D2 receptor antagonists
- Sleep
- Stress
Additional Control
- PRL can regulate its own secretion via a short feedback loop
- Secretion is pulsatile
- Levels increase with sleep
- need for surfactant synthesis of fetus

8. Gonadotrophins (FSH & LH)
Chemical Nature
- Glycoproteins
- Molecular weight ~30 000 Daltons
- Structure:
- Common α-subunit
- Hormone-specific β-subunit (confers biological specificity)
➡️ hCG belongs to same hormone family.
Cells of Origin
- Secreted by gonadotrophs
- Gonadotrophs = 10–15% of anterior pituitary cells
Receptors & Signalling
- Receptors:
- G-protein-coupled receptors
- Binding activates:
- Adenylate cyclase
- ↑ cAMP
Physiological Actions
Testis
- LH → Leydig cells (interstitial cells)
- FSH → Sertoli cells
Ovary
- Both hormones act on multiple cell types
➡️ Central role in steroidogenesis

Isoforms & Clinical Measurement
- Multiple circulating isoforms of LH & FSH
- Biological potency depends on:
- Degree of glycosylation
- Sites of glycosylation
- Electrical charge
➡️ Consequence:
- Assay concentration ≠ true biological activity in all cases
Final Logic Lock (Integrated One-Sentence Concept)
The anterior pituitary is a glandular endocrine organ regulated by hypothalamic releasing and inhibitory hormones, peripheral feedback, and local autocrine/paracrine signals, producing six key hormones whose secretion patterns, molecular structures, signalling pathways, and feedback controls are precisely adapted to growth, metabolism, reproduction, and lactation.
POSTERIOR PITUITARY GLAND (NEUROHYPOPHYSIS)
Hormones
- Secretes arginine vasopressin (AVP) and oxytocin (OXY).
- AVP and OXY are small peptides with strong structural homology.
- Each consists of 9 amino acids arranged in a ring structure with a short tail.
Sites of Synthesis
- AVP:
- Mainly synthesised in supraoptic nuclei.
- To a lesser extent in paraventricular nuclei.
- OXY:
- Mainly synthesised in paraventricular nuclei.
- To a lesser extent in supraoptic nuclei.
Neuronal Types
- Magnocellular neurosecretory neurons:
- Synthesise AVP and OXY.
- Hormones are transported to and released from the posterior pituitary.
- Parvocellular neurons:
- Control anterior pituitary hormone secretion.
Hormone Synthesis and Transport
- AVP and OXY are synthesised as large prohormones.
- Prohormones are packaged into secretory granules.
- Granules are transported by axonal flow to nerve terminals (Herring bodies) in the neurohypophysis.
- During transport, prohormones are cleaved into:
- Biologically active hormone (AVP or OXY).
- A larger polypeptide fragment (neurophysin).
Neurophysins
- Neurophysin II:
- Cleaved from vasopressin prohormone.
- Neurophysin I:
- Cleaved from oxytocin prohormone.(OXY_PARA-1)
- Neurophysins are co-secreted with AVP and OXY during electrical activation of neurons.
ARGININE VASOPRESSIN (AVP)
Nomenclature
- Named for its pressor effect (raises blood pressure).
- Also called antidiuretic hormone (ADH) due to renal actions.
- AVP and ADH are used interchangeably.
Receptors and General Role
- AVP has three known receptors.
- Acts with aldosterone and atrial natriuretic peptide to regulate blood volume and pressure.
Renal Actions
- Acts on V2 receptors (G-protein linked).
- Receptors located on capillary (basal) side of:
- Distal convoluted tubules
- Collecting ducts
- V2 receptor activation → ↑ cAMP.
- cAMP activates a kinase on the luminal (apical) membrane.
- Leads to insertion of aquaporin water channels into luminal membrane.
- Water moves:
- Into tubular cells
- Across basal membrane
- Into interstitium
- Back into circulation
Osmotic Gradient
- Cortex → medulla osmotic gradient created by loop of Henle counter-current mechanism.
- Collecting ducts pass through this gradient.
- Increasing amounts of solute-free water are reabsorbed.
- This process is controlled by aquaporins, and therefore by AVP.
Regulation of AVP Secretion
Osmolality
- ↑ Extracellular fluid osmolality → ↑ AVP release.
- Detected by osmoreceptors located in:
- Hypothalamus
- Circumventricular organs
- Systemic viscera
- Osmoreceptors also stimulate thirst.
- Net effect:
- ↑ Water retention
- ↓ Serum osmolality
Blood Volume and Pressure
- AVP secretion stimulated by 5–10% reduction in effective blood volume.
- Detected by stretch receptors:
- Baroreceptors
- Other cardiovascular receptors
- Example:
- Haemorrhage → ↑ AVP → ↑ water retention → ↑ blood volume
- Increased volume/pressure → ↓ AVP secretion
Other Stimuli
- Emotional stress
- Pain
- Drugs
- Nausea and vomiting (very potent)
- Important in postoperative water balance
Inhibition
- Alcohol strongly inhibits AVP release.
- As little as 30–90 ml of whiskey can cause inhibition.
- Results in:
- Water loss
- Dehydration
- Hangover symptoms
Vascular Actions
- AVP causes vasoconstriction of arteriolar smooth muscle.
- Mediated via:
- Calcium
- Phospholipase-C second-messenger pathway
- Important in maintaining blood pressure after haemorrhage.
Endocrine Interaction
- AVP potentiates CRH action on pituitary corticotrophs.
OXYTOCIN (OXY)

- Peptide hormone
- Nonapeptide → 9 amino acids
- Synthesized in hypothalamus
- Released from posterior pituitary
General Characteristics
- Neuropeptide acting as:
- Neurohormone
- Neurotransmitter
- Neuromodulator
- Produced centrally in magnocellular hypothalamic neurons.
Peripheral Production
- Also synthesised in:
- Uterus
- Placenta
- Amnion
- Corpus luteum
- Testis
- Heart
Receptor Distribution
- OXY receptors found in:
- Kidney
- Heart
- Thymus
- Pancreas
- Adipocytes
Physiological Actions
Lactation
- Major action in humans.
- Neuroendocrine reflex during suckling.
- Causes contraction of myoepithelial cells around mammary alveoli.
- Leads to milk let-down.
Parturition
- Major role in animals.
- In humans:
- Less evidence for primary role
- Induces powerful uterine contractions
- Synthetic OXY used:
- To induce labour
- To reduce postpartum haemorrhage
Renal Effects
- Can stimulate AVP V2 receptors.
- High-dose IV OXY (in 5% dextrose) can cause:
- Water retention
- Iatrogenic hyponatraemia
- No naturally occurring disease due to excess OXY.
GnRH (Gonadotropin-Releasing Hormone) from hypothalamus
Origin & Nature
- Decapeptide hormone
- Synthesized in hypothalamus
- Mainly from arcuate nucleus
- Secreted into portal hypophyseal circulation
- Acts on gonadotrophs of anterior pituitary
Secretion Pattern
- Secreted in a pulsatile manner ✅ (ESSENTIAL)
- Pulse generator located in hypothalamus
- Frequency and amplitude determine biological effect
- Continuous (non-pulsatile) GnRH →
- Down-regulation of GnRH receptors
- ↓ FSH & LH secretion
➕ Added
- Pulsatility is mandatory for normal gonadotropin secretion
- Loss of pulsatility → hypogonadotropic hypogonadism
Neuroendocrine Control (KNDY Neurons)
- Kisspeptin:
- Directly stimulates GnRH neurons
- Drives pulsatile GnRH secretion
- KNDY neurons (Kisspeptin, Neurokinin B, Dynorphin):
- Coordinate pulse generation
- Estrogen negative feedback:
- Suppresses kisspeptin release
- Some KNDY neurons provide inhibitory signals → ↓ pulse frequency
➕ Added
- Neurokinin B → stimulates GnRH pulses
- Dynorphin → inhibits GnRH pulses
Hormonal Feedback Regulation
- Estrogen:
- ↑ GnRH pulse frequency
- Progesterone:
- ↓ GnRH pulse frequency
- During luteal phase:
- Progesterone dominance → slow pulses
- End of luteal phase:
- ↓ Estrogen & progesterone → pulse frequency rises again
➕ Added
- Estrogen has dual feedback:
- Negative feedback most of cycle
- Positive feedback at mid-cycle → LH surge
Physiologic Effects of Pulse Frequency
- High-frequency pulses → ↑ LH secretion
- Low-frequency pulses → favor FSH secretion
➕ Added
- Differential gene transcription of LH β and FSH β subunits depends on pulse pattern
Ovulatory Phase Changes
- At ovulation:
- Gonadotrophs show self-priming
- ↑ sensitivity to GnRH
- Leads to LH surge
Neurotransmitter Modulation
- Pulse generator:
- Stimulated by:
- Epinephrine (EP)
- Norepinephrine (NEP)
- Inhibited by:
- Enkephalins
- β-endorphins
➕ Added
- Stress, weight loss, excessive exercise → ↑ β-endorphins → ↓ GnRH
Action at Pituitary
- GnRH binds to GnRH receptors (G-protein coupled)
- Stimulates release of:
- LH (more prominent effect)
- FSH
➕ Added
- Acts via IP₃–DAG pathway → Ca²⁺ influx
- Promotes synthesis + release of gonadotropins
Clinical Correlation (Very High Yield)
➕ Added
- GnRH agonists (continuous) → medical castration
- Endometriosis
- Fibroids
- Prostate cancer
- Pulsatile GnRH therapy:
- Treats hypothalamic amenorrhea
- Kisspeptin deficiency → delayed puberty
Used in:
EXAM REFLEX BLOCK — Posterior Pituitary (Neurohypophysis) + GnRH Axis 🧠🎯
Use this as final-hour consolidation. Every line is examiner-triggered.
POSTERIOR PITUITARY (NEUROHYPOPHYSIS)
Core Identity
- Does NOT synthesise hormones → only stores & releases.
- Hormones made in hypothalamic magnocellular neurons.
- Hormones:
- AVP (ADH)
- Oxytocin
- Both are nonapeptides (9 AAs) with ring + tail, high homology.
Sites of Synthesis (Classic Pairing)
- AVP → mainly supraoptic nucleus
- Oxytocin → mainly paraventricular nucleus
- Minor cross-synthesis exists → examiners love “mainly vs partly”.
Transport & Storage
- Synthesised as large prohormones.
- Packaged in secretory granules.
- Transported by axonal flow down pituitary stalk.
- Stored in Herring bodies.
- During transport → cleaved into:
- Active hormone
- Neurophysin
Neurophysins (Very Tested)
- Neurophysin I → with oxytocin
- Neurophysin II → with vasopressin
- Co-secreted during neuronal depolarisation.
- Function: carrier + stabilisation, not hormonal action.
ARGININE VASOPRESSIN (AVP / ADH)
Names = Functions
- ADH → renal water retention
- Vasopressin → vasoconstriction
Renal Action (V2 Pathway – Gold Standard)
- Receptor: V2 (Gs-coupled).
- Location: basolateral membrane of:
- DCT
- Collecting ducts
- Mechanism:
- ↑ cAMP → protein kinase
- Inserts aquaporin-2 into luminal membrane
- Result:
- ↑ water reabsorption
- ↓ serum osmolality
👉 Key line: AVP controls water reabsorption, not sodium.
Why AVP Works (Osmotic Gradient)
- Cortico-medullary gradient created by:
- Loop of Henle counter-current system
- Collecting ducts pass through gradient.
- AVP decides how much water follows.
Regulation of AVP Secretion
1. Osmolality (Most Sensitive)
- ↑ plasma osmolality → ↑ AVP
- Detected by osmoreceptors:
- Hypothalamus
- Circumventricular organs
- Also stimulates thirst.
2. Volume / Pressure
- Requires 5–10% fall (less sensitive).
- Detected by baroreceptors.
- Example:
- Haemorrhage → ↑ AVP → water retention → BP support.
3. Other Powerful Stimuli
- Pain
- Stress
- Drugs
- Nausea & vomiting (very potent)
- Post-operative state → water retention.
Inhibition
- Alcohol → strong AVP suppression.
- Leads to:
- Diuresis
- Dehydration
- Hangover
Vascular Action
- Acts on V1 receptors.
- Uses PLC → Ca²⁺ pathway.
- Causes arteriolar vasoconstriction.
- Critical in shock / haemorrhage.
Endocrine Interaction
- AVP potentiates CRH → ↑ ACTH release.
OXYTOCIN (OXY)
Nature
- Acts as:
- Neurohormone
- Neurotransmitter
- Neuromodulator
- Produced in magnocellular neurons.
Peripheral Synthesis (Often Forgotten)
- Uterus
- Placenta
- Amnion
- Corpus luteum
- Testis
- Heart
Physiological Actions
1. Lactation (Most Important in Humans)
- Trigger: suckling
- Reflex:
- Nipple → hypothalamus → posterior pituitary
- Action:
- Contraction of myoepithelial cells
- Result:
- Milk ejection (let-down)
👉 Milk production = prolactin, not oxytocin.
2. Parturition
- Strong role in animals.
- In humans:
- Not primary initiator
- Causes powerful uterine contractions
- Synthetic oxytocin:
- Labour induction
- Prevention/treatment of postpartum haemorrhage
Renal Effect (Exam Trap)
- High-dose IV oxytocin:
- Stimulates V2 receptors
- Causes water retention
- Can lead to iatrogenic hyponatraemia
- No natural disease due to oxytocin excess.
GnRH — FINAL EXAM CORE
Essential Rules
- Decapeptide
- Secreted into portal circulation
- Acts on gonadotrophs
- Must be pulsatile ⚠️
Pulse Logic
- Pulsatile GnRH → ↑ LH & FSH
- Continuous GnRH → receptor down-regulation → ↓ LH/FSH
- Loss of pulsatility → hypogonadotropic hypogonadism
Pulse Frequency Effects
- Fast pulses → LH dominant
- Slow pulses → FSH dominant
KNDY Neurons (Modern Exam Favourite)
- Kisspeptin → stimulates GnRH
- Neurokinin B → ↑ pulse generation
- Dynorphin → ↓ pulse frequency
- Estrogen suppresses kisspeptin (negative feedback).
Ovulation Logic
- Sustained estrogen → positive feedback
- GnRH self-priming at pituitary
- → LH surge
Clinical Locks
- Continuous GnRH agonists → medical castration
- Endometriosis
- Fibroids
- Prostate cancer
- Pulsatile GnRH → hypothalamic amenorrhoea
- Kisspeptin deficiency → delayed puberty
ONE-LINE MASTER LOCK
Posterior pituitary stores AVP and oxytocin made in hypothalamic magnocellular neurons; AVP regulates water balance via V2-mediated aquaporin insertion and supports BP via V1 vasoconstriction, while oxytocin mediates milk ejection and uterine contraction; GnRH must be pulsatile to sustain LH/FSH secretion, with frequency determining hormonal dominance.
HYPOTHALAMIC–PITUITARY CONTROL OF PUBERTY
Definition of Puberty
- Stage of physical maturation enabling sexual reproduction.
- Associated with:
- Increased growth rate
- Skeletal changes (e.g. ↑ hip width in girls)
- Increased fat and muscle
- Psychological changes
Physiology of Puberty
Central Initiation
- Puberty begins with ↑ GnRH secretion.
- GnRH neurons located in:
- Arcuate nucleus
- Other hypothalamic nuclei
- GnRH drives ↑ LH and FSH secretion.
Pre-Pubertal State
- LH and FSH secreted in very small amounts.
- Low peripheral concentrations.
- No significant gonadal stimulation.
Pubertal Transition
- ↑ Amplitude of pulsatile LH and FSH secretion.
- ↑ Mean secretion rates.
- Nocturnal rise in LH secretion becomes amplified.
- This nocturnal rhythm:
- Specific to puberty
- Disappears in adulthood
Central Programming
- Gonads not required for initiation.
- Brain is programmed to:
- Increase GnRH output
- Increase LH and FSH secretion
Peripheral Effects
- ↑ LH and FSH → gonadal maturation.
- ↑ Sex steroid secretion.
- Along with adrenal androgens, cause pubertal physical changes.
Sex-Specific Changes
Boys
- Earliest sign:
- ↑ Testicular size
- Volume > 4 ml or
- Length > 2.5 cm
Girls
- Thelarche (breast development) indicates ovarian steroid secretion.
- Estradiol secretion fluctuates widely before menarche.
- Reflects waves of follicular development without ovulation.
- Estrogen stimulates uterine growth.
- Menarche occurs when:
- Sufficient uterine growth achieved
- Estrogen withdrawal triggers first menstruation
- Primary amenorrhoea indicates underlying pathology.
Adrenarche and Gonadarche
- Axillary and pubic hair growth due to:
- Gonadal steroids (gonadarche)
- Adrenal steroids (adrenarche)
EXAM REFLEX BLOCK — Hypothalamic–Pituitary Control of Puberty 🧠🎯
Use this as your rapid-fire recall + examiner trap shield.
Core Trigger (Always start here)
- Puberty is centrally initiated by ↑ GnRH pulsatility.
- GnRH neurons: mainly arcuate nucleus (+ other hypothalamic nuclei).
- Leads to ↑ LH & FSH amplitude and mean secretion.
Pre-puberty vs Puberty (Classic Contrast)
- Pre-puberty:
- LH/FSH → very low, minimal pulses.
- Gonads inactive despite being structurally normal.
- Puberty:
- ↑ pulse amplitude, not frequency alone.
- Nocturnal LH rise appears → puberty-specific, gone in adults.
👉 Exam trap: If they ask “what changes first?” → Amplitude of pulsatile secretion.
Central Programming (High-Yield Concept)
- Gonads NOT required to start puberty.
- Brain has an intrinsic developmental clock.
- Peripheral sex steroids are effects, not triggers.
👉 Question stem with gonadal failure + pubertal LH rise = central drive intact.
Peripheral Execution
- LH/FSH → gonadal maturation.
- ↑ Sex steroids + adrenal androgens → physical pubertal changes.
- Adrenal contribution explains hair development independent of gonads.
Sex-Specific Exam Locks
Boys
- Earliest sign of puberty:
- ↑ testicular size
- Volume > 4 mL or length > 2.5 cm
- NOT voice change, NOT pubic hair.
👉 MCQ favourite: “first sign of puberty in boys?” → testicular enlargement.
Girls
- Thelarche = first clear sign → estrogen effect.
- Estradiol secretion:
- Highly fluctuating
- Follicular waves without ovulation.
- Menarche:
- Requires adequate uterine growth.
- Occurs due to estrogen withdrawal, not estrogen peak.
👉 Primary amenorrhoea = always pathological → investigate.
Adrenarche vs Gonadarche (Very Tested Pair)
- Adrenarche:
- Adrenal androgens (DHEA, DHEAS).
- Causes axillary & pubic hair.
- Gonadarche:
- Gonadal sex steroids.
- Responsible for true sexual maturation.
👉 Hair development ≠ proof of puberty onset.
One-Line Exam Reflex
Puberty is centrally programmed by increased GnRH pulsatility (↑ amplitude), producing a transient nocturnal LH rise, leading to gonadal steroid secretion; gonads execute puberty but do not initiate it.
DISORDERS OF PUBERTY, AMENORRHOEA & HYPOTHALAMO–PITUITARY DISEASE
(Logic-based, no omissions)
1. NORMAL PUBERTY → KEY CONCEPT: CONSONANCE
- Puberty follows a reliable, ordered sequence of physical and hormonal changes.
- This coordinated progression is termed consonance.
- Disorders of puberty represent early, delayed, or disordered activation of this sequence.
2. PRECOCIOUS PUBERTY
Definition
- Girls: Onset of puberty before 8 years
- Boys: Onset of puberty before 9 years
⚠️ Important epidemiological update:
- Early pubertal signs (breast development, pubic hair) are commonly seen in girls aged 6–8 years, especially Black girls.
A. CENTRAL PRECOCIOUS PUBERTY (CPP)
= Gonadotropin-dependent
Core mechanism
- Early maturation of the entire HPG axis
- Normal pubertal sequence, but too early
Features
- Full spectrum of:
- Physical pubertal changes
- Hormonal pubertal changes
- Puberty progresses normally but prematurely
Causes
- Idiopathic in:
- ~90% of girls
- Can be associated with:
- CNS tumours
- Brain injury
- Congenital brain anomalies
B. PERIPHERAL PRECOCIOUS PUBERTY (PSEUDOPUBERTY)
= Gonadotropin-independent
Core mechanism
- Excess sex steroid production
- No activation of the HPG axis
Key point
- Much less common than central precocious puberty
3. DELAYED PUBERTY
Definitions
- Boys: Testicular volume < 4 mL by age 14
- Girls: No breast development by 13–15 years
Constitutional Delay
- Most common cause
- Much more frequent in boys
- Affects growth and puberty together
- Investigations are normal by definition
Causes of Delayed Puberty (Table 16.1 – fully preserved)
A. CONSTITUTIONAL DELAY
- Common (~90%)
B. HYPOGONADOTROPHIC HYPOGONADISM (~10%)
- GnRH deficiency
- May be isolated
- Or associated with:
- Anosmia (Kallmann syndrome)
- Cognitive impairment
- Dysmorphic features (e.g. Prader–Willi syndrome)
- Gonadotrophin deficiency
- Isolated:
- Fertile eunuch syndrome (LH deficiency)
- More commonly:
- Associated with hypopituitarism
C. HYPERGONADOTROPHIC HYPOGONADISM (~10%)
- Sex chromosome abnormalities:
- Boys: Klinefelter syndrome (47,XXY)
- Girls: Turner syndrome (45,X)
- Gonadal dysgenesis with normal karyotype
- Gonadal damage:
- Viral (e.g. mumps orchitis)
- Iatrogenic (surgery, chemotherapy, radiotherapy)
- Autoimmune (often with Addison’s disease)
- Loss-of-function mutation:
- LH β-subunit
- Reduced bioactivity
- Elevated LH on immunoassay
- Gonadotrophin receptors
- Resistant ovary syndrome
4. AMENORRHOEA
Definition
- Absence of menstruation in a woman of reproductive age
Physiological amenorrhoea
- Pregnancy
- Lactation
- Childhood
- Post-menopause
Types
Primary amenorrhoea
- No menarche by 16 years
- Can be a feature of delayed puberty
Secondary amenorrhoea
- Absence of menstruation for ≥3 months
- In a previously menstruating woman
CRITICAL CLINICAL LOGIC: BREASTS + UTERUS
Breast development | Uterus | Likely pathology |
Absent | Present | Failure of entire HPO axis (e.g. Kallmann, ovarian failure before puberty) |
Present | Present | Axis failure after breast development |
Present | Absent | Androgen insensitivity or congenital absence of uterus |
Endocrine classification
- High LH/FSH → Primary ovarian failure
- Low LH/FSH → Hypothalamo–pituitary dysfunction
5. DISEASES OF THE HYPOTHALAMUS & PITUITARY
General principles
- Disease manifests as:
- Hormone deficiency
- Hormone excess
- Hormone excess:
- Usually due to pituitary adenoma
- Typically single-hormone over-secretion
- Pituitary tumours:
- Mostly benign adenomas
- May compress optic chiasm
- Carcinomas are rare, aggressive, invasive
Axis terminology
- Primary disorder: End-organ disease
- Secondary disorder: Pituitary dysfunction
- Tertiary disorder: Hypothalamic dysfunction
🧠 EXAM REFLEX BLOCK — Disorders of Puberty, Amenorrhoea & HPO Axis
Use this as instant recall logic in SBAs, vivas, and structured essays.
1️⃣ PUBERTY — FIRST REFLEX
Ask: Is the sequence normal but mistimed, or disordered?
- Normal order, early → Central precocious puberty
- Isolated / mismatched features → Peripheral precocious puberty
- Slow child + late puberty → Constitutional delay
👉 Keyword: Consonance = ordered, coordinated pubertal progression.
2️⃣ PRECOCIOUS PUBERTY — CORE SPLIT
Age cut-offs
- Girls < 8 yrs
- Boys < 9 yrs
Central (GnRH-dependent)
- Entire HPG axis switched on early
- Normal pubertal sequence
- Idiopathic in ~90% of girls
- Think CNS causes if boy or neurological signs
Peripheral (GnRH-independent)
- Sex steroids ↑ without HPG activation
- Puberty not consonant
- Much less common
3️⃣ DELAYED PUBERTY — FIRST DIAGNOSIS IS…
➡️ Constitutional delay (especially boys)
Definitions
- Boys: testes < 4 mL at 14 yrs
- Girls: no breast development by 13–15 yrs
Three-way split
- Constitutional delay (~90%)
- Hypogonadotrophic hypogonadism (central problem)
- Hypergonadotrophic hypogonadism (gonadal failure)
4️⃣ DELAYED PUBERTY — EXAM PATTERNS
Low LH/FSH (Hypogonadotrophic hypogonadism (central problem))
- GnRH deficiency
- Kallmann = delayed puberty + anosmia
- Often part of hypopituitarism
High LH/FSH (Hypergonadotrophic hypogonadism (gonadal failure))
- Gonadal failure/dysgenesis
- Turner (girls), Klinefelter (boys)
- Gonadal damage (mumps, chemo, auto-immune)
- Receptor defects → hormones high but ineffective
5️⃣ AMENORRHOEA — ALWAYS START HERE
Physiology first
- Pregnancy, lactation, childhood, menopause
Definitions
- Primary: no menarche by 16 yrs
- Secondary: no periods ≥3 months
6️⃣ AMENORRHOEA — GOLD TABLE REFLEX
Breasts + Uterus = localisation
- ❌ Breasts + ✅ Uterus → Axis failure before puberty
- ✅ Breasts + ✅ Uterus → Axis failure after puberty
- ✅ Breasts + ❌ Uterus → Androgen insensitivity / Müllerian agenesis
This table alone answers multiple MCQs.
7️⃣ AMENORRHOEA — HORMONAL LOGIC
- High LH/FSH → Ovarian failure
- Low LH/FSH → Hypothalamo-pituitary cause
Never miss this step.
8️⃣ HYPOTHALAMO–PITUITARY DISEASE — EXAM RULES
- Think deficiency OR excess
- Excess = usually single hormone adenoma
- Most tumours = benign
- Visual field loss = optic chiasm compression
- Carcinoma = rare, aggressive
9️⃣ AXIS TERMINOLOGY — MUST-USE WORDS
- Primary → end-organ problem
- Secondary → pituitary problem
- Tertiary → hypothalamic problem
Examiners expect this language.
🔒 FINAL MEMORY LOCK
If confused in exam:
- Age
- Sequence (consonant or not)
- Breasts + uterus
- LH/FSH direction
- Primary vs secondary vs tertiary
6. ACROMEGALY
Definition
- Chronic exposure to excess GH
Epidemiology
- Incidence: ~3/million/year
- Onset: 3rd–4th decade
- Equal in both sexes
- Delay to diagnosis: 5–10 years
Causes
- 95%: GH-secreting pituitary adenomas
- Often macroadenomas (>1 cm)
- 15%: Co-secrete PRL
- Rare causes:
- Hypothalamic tumours (glioma, hamartoma)
- Peripheral tumours (pancreatic adenocarcinoma)
Pathophysiology
- GH secretion:
- Still pulsatile
- Increased amplitude, duration, frequency
- Absent nocturnal surge
- Abnormal suppression & stimulation responses
- Effects mediated mainly by IGF-I
- Bone, cartilage, soft tissue proliferation
- Organ enlargement
Pregnancy physiology
- Placenta produces:
- Variant GH
- GH-releasing hormone
- IGF-I
- Placental GH:
- Rises through pregnancy
- Falls rapidly after delivery
Clinical features (Table 16.2 – preserved)
Skeletal
- Arthralgia/arthritis (85%)
- Carpal tunnel (50%)
- Enlarged hands/feet (100%)
- Jaw protrusion (90%)
- Dental malocclusion (80%)
- Osteoarthritis (30%)
Skin
- Excessive sweating (85%)
- Greasy skin, skin tags (60%)
Cardiovascular
- Angina (5–10%)
- Hypertension (50%)
- Cardiomyopathy (5%)
Respiratory
- Daytime somnolence (30%)
- Obstructive sleep apnoea (30%)
Metabolic
- Polydipsia/polyuria (5%)
- Neuropathy (50%)
- Retinopathy (15%)
Renal
- Renal colic (20%)
- Renal stones (20%)
Endocrine
- Menstrual irregularity (50%)
- Impotence (40%)
- Hypogonadism (40%)
Mortality
- Cardiovascular/cerebrovascular: 36–62%
- Respiratory: 0–25%
- Malignancy: 9–25%
- Higher if diabetes or hypertension present
Diagnosis
- Oral glucose tolerance test
- 75 g glucose
- Normal: GH < 2 mU/L
- Acromegaly:
- Failure to suppress
- May show paradoxical increase
- TRH or GnRH stimulation:
- GH rises in 70–80%
- Visual field testing:
- Bitemporal hemianopia
- Imaging:
- MRI – modality of choice
Treatment
- Surgery (best if tumour <10 mm)
- Medical therapy
- Radiotherapy
- Panhypopituitarism in 15–20% after years
🧠 EXAM RECALL BLOCK — ACROMEGALY
Definition
- Chronic exposure to excess growth hormone (GH).
Epidemiology
- Incidence ~3/million/year.
- Onset 3rd–4th decade.
- Equal sex distribution.
- Diagnosis delayed 5–10 years.
Cause
- ~95%: GH-secreting pituitary adenoma.
- Usually macroadenoma (>1 cm).
- ~15% co-secrete prolactin.
- Rare: hypothalamic tumours, ectopic GH/GHRH (e.g. pancreatic).
Pathophysiology
- GH secretion remains pulsatile.
- ↑ Amplitude, duration, frequency.
- Absent nocturnal surge.
- Failure of suppression with glucose.
- Effects mediated mainly by IGF-I → bone, cartilage, soft-tissue, organ overgrowth.
Pregnancy physiology
- Placenta produces variant GH, GHRH, IGF-I.
- Placental GH rises during pregnancy, falls rapidly post-delivery.
Clinical features
- Skeletal: enlarged hands/feet (100%), prognathism (90%), malocclusion (80%), arthralgia (85%), carpal tunnel (50%).
- Skin: sweating (85%), greasy skin/skin tags (60%).
- CVS: hypertension (50%), cardiomyopathy (5%), angina (5–10%).
- Respiratory: OSA + daytime somnolence (~30%).
- Metabolic/neurologic: neuropathy (50%), PU/PD (5%), retinopathy (15%).
- Renal: renal colic/stones (20%).
- Endocrine: menstrual irregularity (50%), impotence (40%), hypogonadism (40%).
Mortality
- Cardio/cerebrovascular: 36–62% (commonest).
- Respiratory: 0–25%.
- Malignancy: 9–25%.
- Risk ↑ with diabetes or hypertension.
Diagnosis
- OGTT (75 g glucose):
- Normal: GH < 2 mU/L.
- Acromegaly: fails to suppress Gh ± paradoxical rise.
- TRH or GnRH stimulation: GH rises in 70–80%.
- Visual fields: bitemporal hemianopia.
- MRI pituitary: investigation of choice.
Treatment
- Transsphenoidal surgery (best if tumour <10 mm).
- Medical therapy (somatostatin analogues, dopamine agonists, GH receptor antagonists).
- Radiotherapy → panhypopituitarism 15–20% (late).
7. GROWTH HORMONE DEFICIENCY
Childhood
- Detected early due to reduced growth velocity
Adults
- Due to hypothalamo–pituitary damage
Diagnosis
- Insulin tolerance test
- Peak GH < 9 mU/L
- Contraindications:
- Seizures
- Ischaemic heart disease
8. HYPERPROLACTINAEMIA
Causes (Table 16.3 – preserved)
Common (~90%)
- Drugs:
- Neuroleptics (phenothiazines)
- Dopamine antagonists (metoclopramide)
- Primary hypothyroidism
Uncommon (~10%)
- Macroprolactinaemia
- Stalk syndrome
- Dopamine interruption
- PRL <3000 mU/L
- Pituitary tumours
- PRL >5000 mU/L
Rare (<1%)
- Renal failure
9. PROLACTINOMA
Most common hormone-secreting pituitary tumour
Classification
- Microprolactinoma: <10 mm
- Macroprolactinoma: >10 mm
Physiology of lactotrophs
- Normally:
- ~20% of pituitary cells
- Pregnancy:
- Up to 50%
- Postpartum:
- Partial regression
- Hyperplasia persists up to 11 months
- Decidual PRL:
- Same as pituitary PRL
- Not inhibited by dopamine
Clinical differences
- Women:
- Smaller tumours
- Galactorrhoea
- Amenorrhoea
- Men:
- Larger tumours
- Subtle hypogonadism
Management
- First-line: Dopamine agonists
- Cabergoline
- Bromocriptine
- Safe in pregnancy
- Surgery / radiotherapy / temozolomide if resistant
Pregnancy risk of enlargement
- Microprolactinoma: 1.3%
- Untreated macroprolactinoma: 23.2%
- Treated macroprolactinoma: 2.8%
10. NON-FUNCTIONING PITUITARY ADENOMAS
- No hormone excess
- Symptoms due to mass effect:
- Headache
- Visual defects
- Cranial nerve palsies
- Hypopituitarism
- Investigations:
- Pituitary function
- Visual fields
- MRI
- Treatment:
- Surgery ± radiotherapy
11. CRANIOPHARYNGIOMA & RATHKE CLEFT CYST
Craniopharyngioma
- Extra-axial
- Squamous epithelial
- Calcified
- Benign histology, malignant behaviour
- Age: usually <20 years
- Symptoms:
- Raised ICP
- Hypopituitarism + DI
- Visual field defects
- Treatment:
- Surgery + radiotherapy
- Hormone replacement
Rathke cleft cyst
- Benign, epithelium-lined
- Intrasellar
- Surgery only if symptomatic
- Approach: trans-sphenoidal
12. DIABETES INSIPIDUS
Central DI
- ↓ AVP secretion
- Causes:
- Hypothalamic osmoreceptors
- Supraoptic/paraventricular nuclei
- Pituitary stalk
- Posterior pituitary lesions rarely permanent
Nephrogenic DI
- Renal resistance to AVP
- Causes:
- CKD
- Lithium
- Hypercalcaemia
- Hypokalaemia
- Tubulointerstitial disease
- Genetic:
- X-linked V2 receptor defect
- Autosomal recessive aquaporin-2 mutation
Treatment
- Desmopressin
- Avoid hyponatraemia
13. LYMPHOCYTIC HYPOPHYSITIS
Nature
- Autoimmune inflammatory disorder
- Pituitary ± stalk
Epidemiology
- Common in:
- Pregnancy
- Postpartum
- Can affect any age/sex
- Associated with autoimmune disease
Presentation
- Mimics pituitary adenoma:
- Headache
- Visual loss (bitemporal hemianopia)
- Hypopituitarism
- Diabetes insipidus
- Diplopia, orbital pain (cavernous sinus)
Classification
- Primary (idiopathic)
- Secondary:
- Sarcoidosis
- TB
- Langerhans cell disease
- Wegener’s
- IgG4-related disease
Treatment
- First-line: High-dose corticosteroids
- Hormone replacement often required
- 72% need lifelong replacement
- Surgery:
- Visual loss
- Failed medical therapy
- Radiotherapy:
- In refractory cases
🧠 EXAM RECALL BLOCK — PITUITARY DISORDERS (7–13)
(Ultra-fast • zero explanation • examiner-triggered recall)
7. GROWTH HORMONE DEFICIENCY
Childhood
- Early detection due to ↓ growth velocity.
Adults
- Due to hypothalamo–pituitary damage.
Diagnosis
- Insulin tolerance test (gold standard).
- Peak GH < 9 mU/L.
Contraindications for insulin tolerance test(must-remember)
- Seizure disorder
- Ischaemic heart disease
8. HYPERPROLACTINAEMIA
Common causes (~90%)
- Drugs:
- Neuroleptics (phenothiazines)
- Dopamine antagonists (metoclopramide)
- Primary hypothyroidism
Uncommon (~10%)
- Macroprolactinaemia
- Stalk syndrome
- Dopamine interruption
- moderately high prolactin
- PRL < 3000 mU/L
- Pituitary tumours
- PRL > 5000 mU/L
Rare (<1%)
- Chronic renal failure
9. PROLACTINOMA
Key fact
- Most common hormone-secreting pituitary tumour.
Size
- Microprolactinoma: < 10 mm
- Macroprolactinoma: > 10 mm
Lactotroph physiology
- Normal: ~20% of pituitary cells
- Pregnancy: ↑ to ~50%
- Postpartum: partial regression, hyperplasia up to 11 months
- Decidual prolactin:
- Same as pituitary PRL
- Not dopamine-inhibited
Sex differences
- Women: smaller tumours, galactorrhoea, amenorrhoea
- Men: larger tumours, subtle hypogonadism
Management
- First-line: dopamine agonists
- Cabergoline
- Bromocriptine
- Safe in pregnancy
- Resistant cases → surgery / radiotherapy / temozolomide
Pregnancy risk of enlargement
- Microprolactinoma: 1.3%
- Untreated macroprolactinoma: 23.2%
- Treated macroprolactinoma: 2.8%
10. NON-FUNCTIONING PITUITARY ADENOMAS
Hormones
- No hormone excess
Symptoms = mass effect
- Headache
- Visual defects
- Cranial nerve palsies
- Hypopituitarism
Investigations
- Pituitary function tests
- Visual fields
- MRI
Treatment
- Surgery ± radiotherapy
11. CRANIOPHARYNGIOMA & RATHKE CLEFT CYST
Craniopharyngioma
- Extra-axial
- Squamous epithelial
- Calcified
- Benign histology, malignant behaviour
- Age: usually < 20 years
Presentation
- Raised ICP
- Hypopituitarism + DI
- Visual field defects
Treatment
- Surgery + radiotherapy
- Hormone replacement
Rathke cleft cyst
- Benign, epithelium-lined
- Intrasellar
- Surgery only if symptomatic
- Trans-sphenoidal approach
12. DIABETES INSIPIDUS
Central DI
- ↓ AVP secretion
- Causes:
- Hypothalamic osmoreceptors
- Supraoptic / paraventricular nuclei
- Pituitary stalk
- Posterior pituitary lesions rarely permanent
Nephrogenic DI
- Renal resistance to AVP
- Causes:
- CKD
- Lithium
- Hypercalcaemia
- Hypokalaemia
- Tubulointerstitial disease
- Genetic:
- X-linked V2 receptor defect
- AR aquaporin-2 mutation
Treatment
- Desmopressin
- Avoid hyponatraemia
13. LYMPHOCYTIC HYPOPHYSITIS
Nature
- Autoimmune inflammatory disorder
- Pituitary ± stalk
Epidemiology
- Common in pregnancy & postpartum
- Any age/sex
- Associated with autoimmune disease
Presentation (adenoma mimic)
- Headache
- Bitemporal hemianopia
- Hypopituitarism
- Diabetes insipidus
- Diplopia, orbital pain (cavernous sinus)
Classification
- Primary (idiopathic)
- Secondary:
- Sarcoidosis
- TB
- Langerhans cell disease
- Wegener’s
- IgG4-related disease
Treatment
- First-line: high-dose corticosteroids
- Hormone replacement common
- 72% need lifelong replacement
- Surgery: visual loss / failed medical therapy
- Radiotherapy: refractory cases
Pituitary diseases in pregnancy + Sheehan + Premature ovarian failure
(Logic-based note, zero omission)
1) Prolactinoma in pregnancy
A. Core endocrine logic (why symptoms happen)
- Excess PRL directly inhibits pulsatile GnRH secretion.
- This causes anovulation/amenorrhoea → infertility.
- Dopamine agonists correct hyperprolactinaemia and restore ovulation in ~90% of women with prolactinoma.
B. Two key pregnancy issues (the exam framework)
Issue 1 — Dopamine agonist exposure in early pregnancy
- Concern: fetal exposure before pregnancy is discovered.
- Principle: limit fetal exposure to dopamine agonists.
- Bromocriptine: seems safe in pregnancy.
- Cabergoline: available data suggest safe, but evidence is from a small number of pregnancies.
- Pergolide + quinagolide: safety data too limited to recommend use in pregnancy.
- Reported heart valve damage with high-dose cabergoline:
- Not demonstrated with cabergoline at hyperprolactinaemia treatment doses.
- Not demonstrated with bromocriptine.
- Not demonstrated with quinagolide (noted as a non-ergot dopamine agonist).
Issue 2 — Effect of pregnancy on the prolactinoma
- Pregnancy stimulates normal lactotrophs → normal pituitary enlargement.
- This does not necessarily mean the adenoma enlarges.
- Prolactinomas becoming symptomatic in pregnancy are uncommon.
- Symptoms suggesting tumour growth:
- Headache
- Visual field changes
- Risk of clinically significant enlargement:
- Microprolactinoma: 1–2%
- Untreated macroprolactinoma: about 20%
- Previously treated macroprolactinoma (surgery/radiation/both): 2–5%
- If a prolactinoma has shrunk on dopamine agonist, then after stopping treatment, there is a reduced chance of symptomatic growth during pregnancy.
C. What to do if symptomatic during pregnancy (management logic)
- If a pregnant woman with prolactinoma becomes symptomatic:
- Restart medical treatment is recommended.
- Symptoms usually regress quickly.
- If not responding to medical treatment OR if visual deterioration continues:
- Transsphenoidal surgery or delivery is an alternative.
- Monitoring in known macroadenoma pregnancy:
- Monthly visual field examinations are important.
- MRI is performed if symptoms of enlargement and/or visual field defects develop.
D. Postpartum / lactation facts (must-know)
- Breastfeeding has not been associated with growth of an underlying prolactinoma.
- After pregnancy:
- Resolution of hyperprolactinaemia and regression of prolactinoma have been reported.
- Idiopathic hyperprolactinaemia is even more likely to resolve after pregnancy.
2) Sheehan syndrome (postpartum pituitary necrosis)
A. Trigger + definition
- Severe haemorrhage, shock, or hypotension during or before parturition can cause postpartum pituitary necrosis (Sheehan syndrome).
- Leads to partial or complete hypopituitarism.
- Now uncommon due to improved obstetric practice.
B. Pathogenesis (stepwise mechanism)
- Pregnancy causes ~50% increase in pituitary volume.
- Sudden BP fall (e.g., postpartum haemorrhage) → hypoperfusion → infarction → ischaemia → cellular damage + oedema.
- Oedema → pituitary swelling.
- More likely when there is a known or unknown pituitary mass.
- Higher risk group noted:
- Type 1 diabetes in pregnancy, especially with pre-existing vascular disease.
C. Which parts are affected (vascular logic)
- Usually anterior pituitary is affected.
- Posterior pituitary + hypothalamus are less vulnerable because they are supplied by:
- Inferior hypophyseal artery
- Circle of Willis
- Some women have impaired AVP secretion → partial or overt diabetes insipidus.
D. Clinical presentation (depends on % destruction)
- Presentation is highly variable.
Severe destruction (95–99% anterior pituitary destroyed)
- Postpartum failure of lactation
- Secondary amenorrhoea
- Loss of axillary and pubic hair
- Genital and breast atrophy
- Increasing signs of:
- Secondary hypothyroidism
- Adrenocortical insufficiency
Less extensive destruction (50–95%)
- Atypical form with loss of one or more hormones.
- Pattern of trophic hormone loss is unpredictable, but invariably results in:
- Hypothyroidism (impaired TSH secretion)
- Hypoadrenalism (impaired ACTH secretion)
Neuropsychiatric + diabetes clue
- Mental disturbances are frequent, sometimes overt psychosis.
- These revert with hormone replacement.
- Women with type 1 diabetes may present with decreasing insulin requirements.
E. Imaging
- MRI or CT is indicated to exclude mass lesions.
- Long-standing Sheehan:
- Sella often empty, filled with CSF.
- Sometimes small remnants of pituitary tissue seen.
F. Treatment + prognosis facts
- Treatment is hormone replacement.
- Sometimes gonadotrophin secretion is preserved → pregnancy is possible.
- Spontaneous recovery from hypopituitarism after postpartum haemorrhage has been reported.
3) Premature ovarian failure (POF)
A. Definitions (get the exam wording right)
- Menopause: defined by the last menstrual period, ends reproductive phase.
- Average menopause age: ~51 years.
- Ovarian failure diagnosis: sex steroid deficiency + elevated gonadotrophins + amenorrhoea.
- Premature ovarian failure (POF):
- Statistically: ovarian failure occurring >2 SD below mean menopause age for reference population.
- Or arbitrarily: before age 40 years.
B. Epidemiology + who it affects
- Can occur before or after menarche.
- Prevalence:
- 1% of women under 40
- 0.1% under 30
- Varies by ethnicity:
- Lower risk in women of oriental origin
- Higher risk in black women than white women
- Frequency in amenorrhoea workups:
- 10–28% of primary amenorrhoea
- 4–18% of secondary amenorrhoea
C. Pathophysiology (what goes wrong)
- POF results from either:
- Follicle dysfunction
- Follicle depletion
- Key contrast with normal menopause:
- ~50% of women with POF have intermittent ovarian function up to 15 years after onset.
- Pregnancy can occur even:
- After a diagnosis of POF
- Even if no follicles are seen on ovarian biopsy
D. Causes (full list preserved)
- In most women: no identifiable aetiology.
- Proposed spontaneous mechanisms:
- Small initial follicle pool
- Inappropriate luteinisation of Graafian follicles
- Other causes:
- Chromosomal/genetic abnormalities (X chromosome or autosomes)
- Autoimmune ovarian damage with positive anti-ovarian antibodies
- Iatrogenic: pelvic surgery, radiotherapy, chemotherapy
- Environmental: viral infections or toxins (no clear mechanism)
E. Clinical presentation (variable patterns)
- Rare presentation:
- Primary amenorrhoea with variable secondary sexual development
- More common:
- Secondary amenorrhoea
- Presentation contexts:
- Symptoms of estrogen deficiency or menstrual disturbance
- Infertility work-up
- Part of genetic/autoimmune syndrome
- Chromosomal defects:
- Often primary amenorrhoea + absent secondary sex characteristics
- Mosaicism:
- Some functioning gonadal tissue
- Variable sexual development + transient menstruation possible
- No specific warning signs of approaching POF.
- “Prodromal POF”:
- ~50% have oligomenorrhoea or dysfunctional uterine bleeding before POF develops
- Acute onset in 25%:
- After pregnancy/delivery
- Or after stopping oral contraceptive pill
- Vasomotor symptoms:
- Only in women with secondary amenorrhoea
- May start even when periods still regular → suggests prodromal POF
- Fertility pattern:
- Usually normal before disorder starts
- Decline may show as resistant ovaries or rising FSH
F. Health risks (long-term consequences)
- Women with POF have nearly twice the age-specific mortality risk.
- Because sex steroid deficiency starts earlier and lasts longer:
- Higher risk of osteoporosis
- Especially high if POF occurs before peak adult bone mass
- Bone density statistic:
- Two-thirds of women with normal karyotype + POF have BMD 1 SD below age-matched mean despite standard hormonal therapy
- Translates to 2.6-fold increased hip fracture risk
- Cardiovascular risk increased:
- Increased mortality documented for:
- Coronary heart disease (OR death 1.29)
- Stroke (OR death 3.07)
- Cancer (OR death 1.83)
- Other causes (OR death 2.14)
4) Autoimmune POF (subset, high-yield)
A. How common + what form is reversible
- Autoimmune mechanisms involved in up to 30% of POF.
- Autoimmune oophoritis:
- Important cause in 10–30%
- Cause of reversible POF
- Can be humoral or cellular
B. Antibodies described (specific list)
- Against steroidogenic enzymes (e.g., 3-hydroxy steroid dehydrogenase)
- Against gonadotrophins and their receptors
- Against corpus luteum
- Against zona pellucida
- Against oocyte
- Specificity for disease is unknown.
C. Lymphocytic oophoritis associations
- Increased peripheral T-lymphocyte activity
- Can be isolated or associated with:
- Addison disease
- Diabetes mellitus
- Myasthenia gravis
- Systemic lupus erythematosus
- Rheumatoid arthritis
- Autoimmune hypothyroidism
- Lymphocytic infiltration may be present in ovarian hilum with lymphocytes accumulating around neural tissue.
D. Autoantibody prevalence facts
- 40% of women with POF have at least one organ-specific autoantibody.
- Most common: antithyroid antibodies (~20%).
5) Genetics, infections, and iatrogenic causes (expanded facts preserved)
Familial risk
- 5–30% have another affected female relative.
- Inheritance could be:
- X-linked
- Autosomal dominant
- Autosomal recessive
- Causal mutation often unknown.
Chromosomal/genetic associations
- Turner syndrome (45,X)
- Triple X syndrome
- Fragile X syndrome
- Fragile X premutation
- Swyer syndrome (pure gonadal dysgenesis with XY constitution)
- Blepharophimosis
- Perrault syndrome
- Down syndrome
Infections linked
- Oophoritis after:
- Mumps
- Malaria
- Varicella
- Shigella infections
Radiotherapy / chemotherapy (dose facts)
- Younger women more resistant; prepubertal ovaries least susceptible.
- Complete ovarian failure:
- 20 Gy in women under 40
- 6 Gy in older women
6) Diagnosis of POF (exam criteria)
Core diagnostic rule
- Elevated serum FSH 40 IU/L
- On at least two occasions
- Separated by a few weeks
Why two samples are required
- Natural history is variable with relapse/remission (“fluctuating ovarian function”).
Pregnancy rate despite diagnosis
- Approximately 1–5% pregnancy rate reported.
Tests that add little / what imaging is for
- Ovarian biopsy:
- Adds little
- Small sample not predictive of natural history
- Pelvic ultrasound:
- Not predictive
- May help identify candidates for oocyte preservation
Additional assessments
- Autoimmune screen:
- Thyroid and adrenal autoantibodies for future surveillance
- Family history:
- Can identify other affected members in up to 30%
Genetic screening facts
- Increasingly used in:
- Familial POF
- Sporadic POF with high suspicion
- Fragile X premutation:
- 15% in women with POF + positive family history
- 3% in sporadic presentations
- Karyotype + fragile X premutation screening should be considered with:
- Family history
- Unusually young onset
7) Management of POF (3 components, preserved)
- Medical treatment
- Fertility advice
- Psychological support
Core medical focus
- Quality of life + bone protection → hormone replacement therapy
Fertility options mentioned
- Oocyte donation
- Adoption
Psychological care
- Personal and emotional support for impact on health and relationships
Follow-up
- Long-term follow-up to:
- Monitor hormone replacement therapy
- Surveillance for emerging autoimmune pathology
🔒 EXAM-REFLEX BLOCK
(Pituitary disease in pregnancy · Sheehan syndrome · Premature ovarian failure)
1️⃣ Prolactinoma in pregnancy — reflex logic
Core reflex
- High PRL → GnRH suppression → anovulation → infertility
- Dopamine agonists restore ovulation (~90%)
Pregnancy risks — remember 2 questions only
- Drug exposure early pregnancy
- Bromocriptine → SAFER
- Cabergoline → probably safe (limited data)
- Pergolide / Quinagolide → avoid
- Valve disease → only high-dose cabergoline (not hyperprolactinaemia doses)
- Tumour growth risk
- Microprolactinoma → 1–2%
- Untreated macro → ~20%
- Treated macro → 2–5%
Symptoms = growth
- Headache
- Visual field defects
Management reflex
- Symptomatic → restart dopamine agonist
- If fails / vision worsening → transsphenoidal surgery OR delivery
- Macroadenoma → monthly visual fields
- MRI → only if symptoms
Post-partum reflex
- Breastfeeding does NOT enlarge prolactinoma
- PRL levels may normalize after pregnancy
- Idiopathic hyperprolactinaemia often resolves
2️⃣ Sheehan syndrome — never miss this
Trigger
- Postpartum haemorrhage / shock / hypotension
Pathogenesis chain (must recall in order)
- Pregnancy → 50% pituitary enlargement wenawa
- ↓ BP → hypoperfusion → infarction → oedema → necrosis
Which part dies?
- Anterior pituitary mainly
- Posterior spared (inferior hypophyseal artery + Circle of Willis)
- DI possible → partial AVP deficiency
Clinical reflex patterns
Severe (95–99% loss):
- Failure of lactation
- Amenorrhoea
- Loss of pubic/axillary hair
- Hypothyroidism + adrenal insufficiency
Partial (50–95%):
- Hormone loss unpredictable
- ALWAYS → ACTH + TSH deficiency
Key exam clues
- Psychosis / mental changes
- Falling insulin needs in type 1 diabetes
Imaging reflex
- Chronic → empty sella (CSF-filled)
- MRI/CT → exclude mass
Treatment pearl
- Hormone replacement
- Gonadotrophins may be preserved → pregnancy possible
- Rare spontaneous recovery exists
3️⃣ Premature ovarian failure (POF) — definition trap area
Definition reflex
- Ovarian failure before 40
- Low estrogen + high FSH + amenorrhoea
- Menopause mean = 51 yrs
Epidemiology numbers (must remember)
- <40 yrs → 1%
- <30 yrs → 0.1%
- Primary amenorrhoea → 10–28%
- Secondary amenorrhoea → 4–18%
Pathophysiology contrast
- Follicle depletion OR dysfunction
- 50% intermittent ovarian activity
- Pregnancy possible even with empty biopsy
4️⃣ Autoimmune POF — reversible subset
Key numbers
- Autoimmune mechanism → up to 30%
- Oophoritis → 10–30%, reversible
Antibodies (recognition list)
- Steroidogenic enzymes
- Gonadotrophins / receptors
- Corpus luteum
- Zona pellucida
- Oocyte
Associations (always test)
- Addison disease
- Type 1 diabetes
- Myasthenia gravis
- SLE
- RA
- Autoimmune hypothyroidism
Autoantibody prevalence
- Any organ-specific antibody → 40%
- Antithyroid → ~20%
5️⃣ Genetics / iatrogenic — exam favourites
Genetic associations
- Turner (45,X)
- Triple X
- Fragile X (premutation)
- Swyer syndrome (XY)
- Perrault
- Blepharophimosis
- Down syndrome
Familial risk
- 5–30%
- X-linked / AD / AR possible
Radiation thresholds
- <40 yrs → 20 Gy
- Older women → 6 Gy
- Prepubertal ovaries → most resistant
6️⃣ Diagnosis of POF — non-negotiable rule
- FSH ≥40 IU/L
- Two samples
- Few weeks apart
Why? → Fluctuating ovarian function
Tests that DON’T help
- Ovarian biopsy → poor predictor
- Pelvic US → not predictive (useful for fertility planning only)
Genetic screening triggers
- Family history
- Very early onset
- Fragile X premutation:
- 15% familial
- 3% sporadic
7️⃣ Management of POF — 3-pillar reflex
- Hormone replacement → QoL + bone protection
- Fertility counselling → oocyte donation / adoption
- Psychological support
Long-term risks (numbers examiners like)
- Mortality × 2
- Hip fracture risk × 2.6
- CHD death OR 1.29
- Stroke death OR 3.07
🧠 FINAL EXAM LOCK
If the question says pregnancy + pituitary → think prolactinoma vs Sheehan.
If it says amenorrhoea + high FSH <40 yrs → POF until proven otherwise.
If it says PPH + no lactation → Sheehan, don’t miss it.